Editor’s Note
This study presents a novel one-pot synthesis of CuBi co-doped mesoporous Beta zeolites for efficient NH₃-SCR denitrification. Comprehensive characterization reveals enhanced catalytic performance, offering a promising route for advanced NOₓ removal materials.
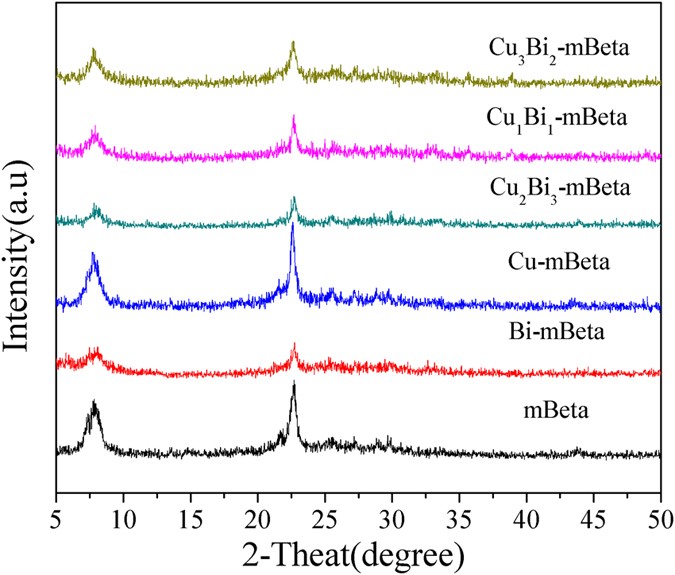
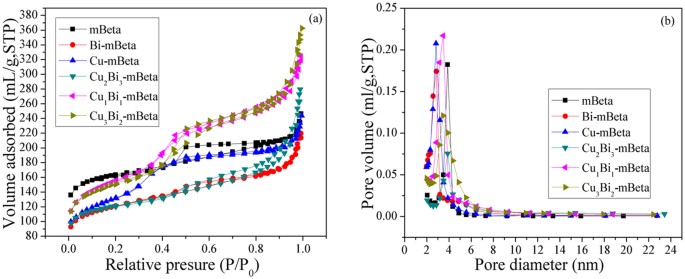
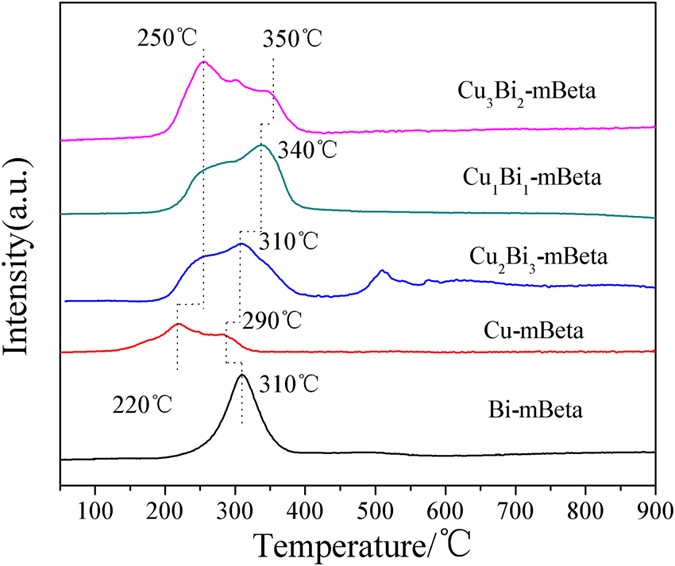
A series of CuBi co-doped mesoporous zeolite Beta (CuxBiy-mBeta) were prepared by a facile one-pot hydrothermal treatment approach and were characterized by XRD, N2 adsorption-desorption, TEM/SEM, XPS, H2-TPR, NH3-TPD and in situ DRIFTS. The catalysts CuxBiy-mBeta were applied to the removal of NOx by selective catalytic reduction with ammonia (NH3-SCR), especially the optimized Cu1Bi1-mBeta achieved the high efficiency for the removal of NOx and N2 selectivity, superior water and sulfur resistance as well as good durability. The excellent catalytic performance could be attributed to the acid sites of the support and the synergistic effect between copper and bismuth species. Moreover, in situ DRIFTS results showed that amides NH2 and NH4+ generated from NH3 adsorption could be responsible for the high selective catalytic reduction of NOx to N2. In addition, a possible catalytic reaction mechanism on Cu1Bi1-mBeta for the removal of NOx by NH3-SCR was proposed for explaining this catalytic process.
Nowadays, it is still of great challenges for the effectively catalytic purification of diesel exhausts, especially for the NOx from diesel engine, since the conventional three-way catalysts are no longer effective in selective reducing NOx1. The commercial selective catalytic reduction with ammonia (NH3-SCR) catalyst for the removal of NOx, i.e., V2O5-WO3/TiO2, only shows high catalytic efficiency in a narrow temperature window of 300–400 °C2, besides, the poor water and sulfur resistance as well as the toxicity of V2O5 also greatly prohibit the popularity of vanadium-based composite oxides. Therefore, researchers have devoted to develop a new kind of non-vanadia catalysts to overcome the disadvantages of vanadium-based composite oxides3. It was reported that compared to the traditional V2O5-WO3/TiO2, non-vanadia catalyst not only presents the wider NH3-SCR temperature windows with high N2 selectivity, but also owns good durability and strong resistance against H2O and SO2.

Very recently, zeolite-based catalysts with high surface areas and pore volume, abundant acidity sites, outstanding thermal and hydrothermal stability, as good catalyst supports, have attracted much research attention in selective catalytic reduction NOx by ammonia4,5. However, small microporous channels of zeolite greatly prevented the diffusion and transport of some large molecules, resulting in the low catalytic performance in a great majority of traditional catalytic reactions6. Therefore, a novel zeolite with mesoporous structure has been developed, which combines the advantages of conventional crystalline zeolite and mesoporus material, to enable the quick access for the diffusion and transport attributed to the hierarchically porous structure. It is generally believed that mesoporous zeolites possess remarkably higher catalytic activity and longer catalytic lifetime than conventional zeolites owing to the crystalline framework and the hierarchically porous structure7. Thereinto, the mesoporous Beta zeolite (mBeta) with unique three-dimensional network of large pores (12MR) exhibits much high surface area and excellent hydrothermal stability, which is widely applied in fine chemistry8. In the past decades, a number of synthetic approaches of mBeta have been explored and some encouraging results have been obtained8,9,10. In addition, many metal oxides were reported to have good performance in the removal of NOx by NH3-SCR, e.g., Cu-loaded zeolite beta exhibited a good activity and hydrothermal stability in the NH3-SCR of NOx11,12. It is also reported that the introduction of Bi2O3 can improve the SO2 resistance13.
On the basis of our previous work14, a novel CuBi co-doped mesoporous zeolite Beta (CuxBiy-mBeta) has been synthesized by one-pot hydrothermal treatment approach, by which the copper and bismuth species can be well dispersed into the framework of mesoporous zeolite Beta. Therein, the optimized prepared catalyst Cu1Bi1-mBeta exhibits very high catalytic activity for the selective catalytic reduction of NOx with NH3. In addition, the N2 selectivity, water vapor and sulfur resistance and durability of the Cu1Bi1-mBeta were also investigated.