Editor’s Note
This article examines the pivotal role of the health sector, including dentistry, in driving the growth and maturity of additive manufacturing. As the industry assesses key players in 2025, the analysis highlights how this field now rivals traditional powerhouses like aerospace and automotive in both impact and market revenue.

To take stock of 2025, it is often necessary to take a critical look at the companies that have consistently kept the sector on alert. One such company is 3D Systems. From technological updates to a leadership positioning in several vertical sectors, 2025 was anything but boring for the additive manufacturing giant.
Even though very little information was communicated at the beginning of the year about 3D Systems’ overall strategy, 2025 ultimately revealed a company that is steadily advancing its additive manufacturing solutions in four key segments:
– Health and Dentistry
– Motorsports, Foundries and Service Bureaus
– Jewelry
– Aerospace, Space and Defense

Digital dentistry remains one of the most established application areas, and 3D Systems’ vast portfolio of integrated solutions continues to help dental labs and clinics produce patient-specific devices.
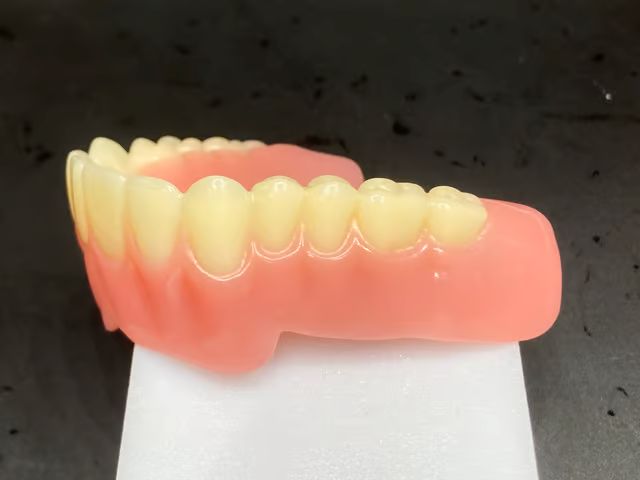
In 2025, the company enhanced its NextDent® Jetted Denture solution for multi-material monolithic dentures, which is now commercially available in the US market. At the heart of this solution is the NextDent® 300 MultiJet 3D printer, a system designed for the rapid production of fully cured, patient-specific dentures that require no additional post-curing steps.
The hardware update is complemented by the introduction of new materials such as NextDent® Jet Teeth and NextDent® Jet Base. These materials are designed for applications such as night guards, a segment experiencing growing demand among dental professionals.
Furthermore, 3D Systems’ clinically validated NextDent 3D printing resin portfolio now covers over 30 applications, including those focused on tooth repair. This includes materials such as NextDent C&B MFH (Micro Filled Hybrid). This material was developed for crowns and bridges, and designed to efficiently produce strong, durable, patient-specific devices.
Beyond dentistry, 3D Systems remains one of the few companies actively advancing biological 3D printing technologies. The company reached a significant milestone this year with FDA clearance for the regenerative repair of peripheral nerve injuries.
This clearance also represents a victory for TISSIUM, the French medical technology company that partnered with 3D Systems to develop a custom 3D printing solution for repairing damaged peripheral nerves.
TISSIUM brings its expertise in the field of programmable biomorphic polymers and 3D Systems brings its bioprinting technologies in regenerative medicine. The result is COAPTIUM® CONNECT with TISSIUM Light, a fully bioabsorbable, 3D-printed medical device that uses a unique photopolymer to promote nerve repair.
That said, as the industry continues to evolve in a “virtuous circle” where applications drive technological progress, we cannot fail to mention the 3D-printed PEEK facial implant, manufactured at the point of care using 3D Systems’ EXT 220 MED.
Produced in collaboration with the University Hospital of Basel (Switzerland), this implant is reported to be the first MDR-compliant facial implant made using Evonik’s VESTAKEEP® i4 3DF PEEK on 3D Systems’ EXT 220 MED and used in a successful surgical procedure performed at the hospital on March 18, 2025.
Search keyword: 3D printed jewelry manufacturing
