Editor’s Note
This article details a significant advance in materials science, where researchers have synthesized diamond particles from a liquid metal alloy at standard atmospheric pressure. This method, requiring far less pressure and lower temperatures than conventional techniques, could pave the way for more accessible industrial and technological applications of synthetic diamonds.

A new method allows for the artificial production of diamonds from a liquid metal alloy without requiring extreme pressure. Conventional techniques require approximately 5 gigapascals of pressure and temperatures around 1400°C. With only 1 atmosphere of pressure (0.0001 gigapascal) and a temperature of 1025°C, the new process initiates the formation of diamond particles within 15 minutes.
In the natural environment, diamonds form through the crystallization of carbon in the Earth’s upper mantle, at depths between 140 and 190 kilometers. At this depth, temperature and pressure are extreme, typically around 1400°C and 4.5-6 gigapascals (GPa). Artificial diamond production methods mimic these conditions using a high-pressure, high-temperature (HPHT) process with carbon-based precursors and alloys.
However, this type of process has limitations, as such high pressures can only be applied in a laboratory on a relatively small surface scale. This implies that the diamonds obtained are generally limited to about one cubic centimeter.
Researchers from the Institute for Basic Science (IBS) in South Korea propose a new method that could enable the production of larger volumes under more accessible conditions. The process, described in the journal *Nature*, has successfully created diamond particles with a pressure of only 1 atmosphere (0.0001 GPa) and a temperature of 1025°C.

To develop their process, the South Korean researchers conducted a series of experiments involving several hundred parameter adjustments. A liquid alloy of gallium, iron, nickel, and silicon was exposed to a gas mixture rich in methane and hydrogen. The setup was placed in a reaction chamber with an internal volume of 100 liters.
However, the formation time of diamond particles was significantly slowed due to the time required to pump air from the chamber (about 3 minutes), purify it with an inert gas (90 minutes), and repump it (3 minutes) until it was completely emptied of all gaseous residue. The next step involves filling the chamber with the purified hydrogen-methane mixture and establishing an internal pressure of 1 atmosphere. Requiring an additional 90 minutes, this final step meant that with this protocol, it would take over 3 hours to initiate diamond production!
To improve the technique and reduce production time, the researchers adjusted the chamber size to 9 liters. This adaptation reduced the pumping and filling time to 15 minutes.
It is noteworthy that the method allows for diamond production without the need for other diamond particles or precursor particles. Indeed, conventional HPHT techniques require the use of precursors to initiate the diamond formation reaction.
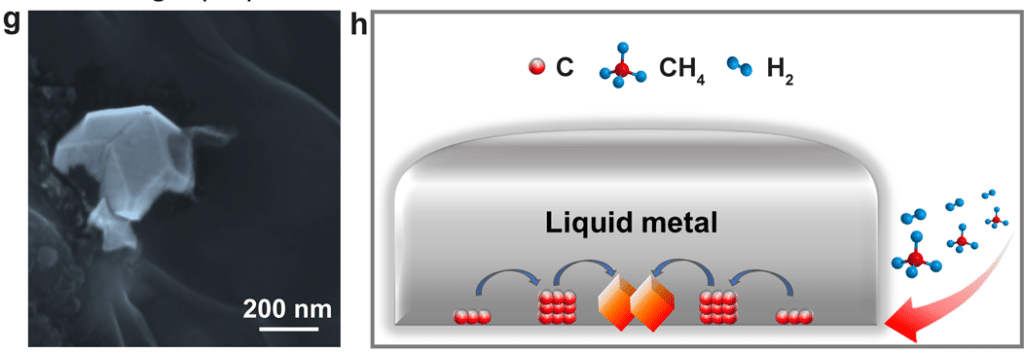
Furthermore, the team discovered that silicon plays an essential role in the size of the final products. If the silicon concentration in the alloy is increased, the size of the resulting diamond is reduced, while its density becomes higher. Silicon appears to promote the formation and stabilization of clusters of carbon atoms that compose the diamond. This suggests that clusters containing silicon atoms could serve as “pre-nuclei,” inducing the formation of diamond particles. The researchers estimate that the size of these initial nuclei is between 20 and 50 carbon atoms.
Moreover, the researchers found that their method offers significant flexibility in the composition of liquid alloys, which is rarely achieved with conventional production techniques. For example, it is possible to use a gallium-nickel-iron-silicon alloy, replace nickel with cobalt, or replace gallium alone with a gallium-indium mixture. Besides methane, a wide variety of gaseous precursors can also be used.
This flexibility would allow for the diversification of the quality and properties of the obtained products, enabling them to be tailored for each specific use.
The gas mixture and liquid alloy are then exposed to a temperature of 1025°C. Once cooled, the solidified alloy exhibits a solid pattern a few millimeters in size that diffracts light into 7 colors, similar to a natural crystal. After analysis, experts found that this crystallized pattern consists of highly purified diamond particles. Once formed, the resulting diamond film can be easily removed and transferred to other substrates for subsequent use.
